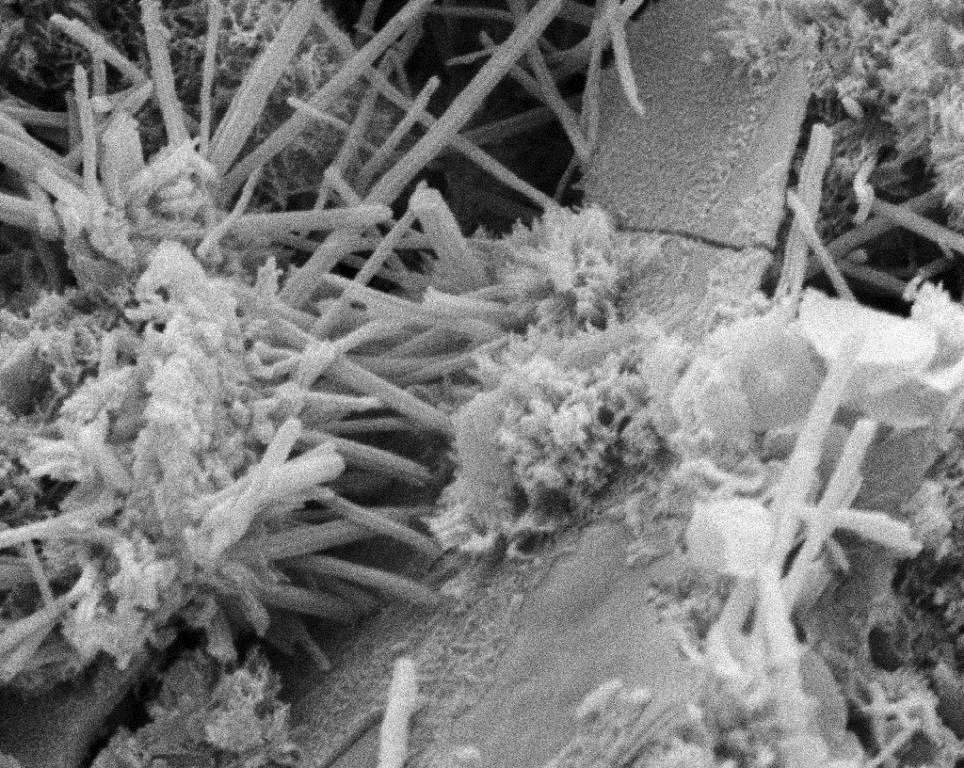
The word “cement” itself is a general word means “a glue” while “portland cement” is a more specific type of glue that is used in the civil engineering construction.
The most important feature of the portland cement is that it is a hydraulic material that reacts with water to give a hard material with strength “paste, mortar, or concrete”. Moreover, if the hardened material was exposed to water over a long time period, beneficial properties such as strength and durability will continue to improve.
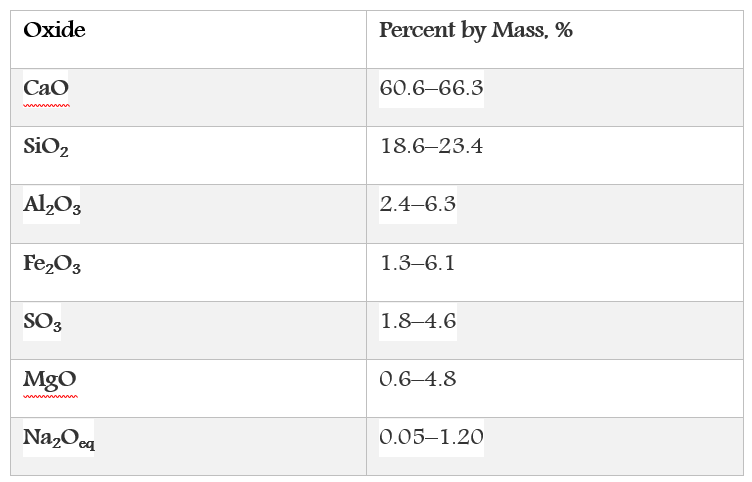
There are four compounds that constitute the portland cement. These compounds are primarily combinations of calcium oxide, silica, alumina, and iron oxide as given in the next table.
The chemical composition of portland cement.
From a practical point of view, the four compounds are considered as follows:
Silicates
- Alite, C3S
- Belite, C2S
Aluminates
- Tricalcium aluminate, C3A
- Calcium alumino ferrite, C4AF
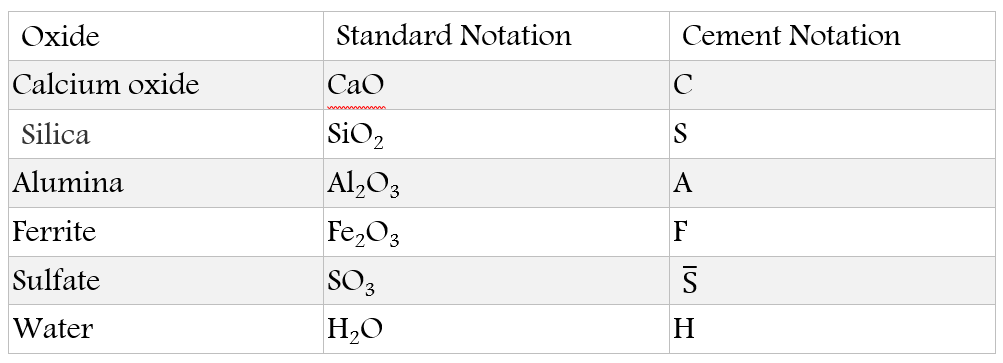
In addition, calcium sulfate compounds (S) are also added to the previous components in the form of gypsum to control the hydration rate of the aluminate phases. Noting that the cement chemistry notation is given as in the following table:
It is important to mention here that the cement compounds are not actually present in the form of oxides, but this approach permits the evaluation of cements according to these numbers. In fact, there is a set of equations “Bouge Equations” available to calculate the Bogue compounds that express the chemistry of the cement as percentages of the aforementioned minerals.
Now, let us take a look on the hydration products of cement. The hydration of the silicates results in different forms of calcium, silica, and water, generically known as calcium-silicate hydrate (C-S-H). C-S-H is responsible for providing the strength and impermeability of concrete. As hydration progresses, the spaces fill in with more C-S-H to form a solid mass. Another hydration product is calcium hydroxide (CH).
When C3A and sulfate hydrates, ettringite is formed at the beginning and then within a few hours converts to monosulfate. When ettringite forms, it can lead to early stiffening and contribute to some of the initial strength.
C4AF does not contribute significantly to the engineering properties of the mixture, but is responsible for the color of cement.